Combining Process Rheology and Spectroscopy Measurements to Improve Batch Polyurethane Polymer Production

In polymer production, the degree of polymerization is strictly dependent on conversion. This very sensitive to impurities and molar imbalance, especially when conversion is high.
Monomer imbalance may prevent the formation of long polymer chains (leading to molecular weight stagnation); and promote the occurrence of undesired secondary reactions (leading to chain branching, gelation and/or chain degradation).
The development and implementation of procedures for online monitoring and control of conversion and weight-average molecular weight (Mw); are highly desirable to avoid gel formation, early Mw stagnation and polymer chain degradation.
Online process measurement tools such as Near-Infrared Process Spectroscopy (NIRS) and Rheology; which includes the measurements of viscosity are both ideal candidates for determining the physical properties of polymers.
Guided Wave and Sofraser have partnered to develop a dual measurement system that can be installed on a single shared flange; which can be installed directly onto the side of a reactor for real-time process control of polymer production.
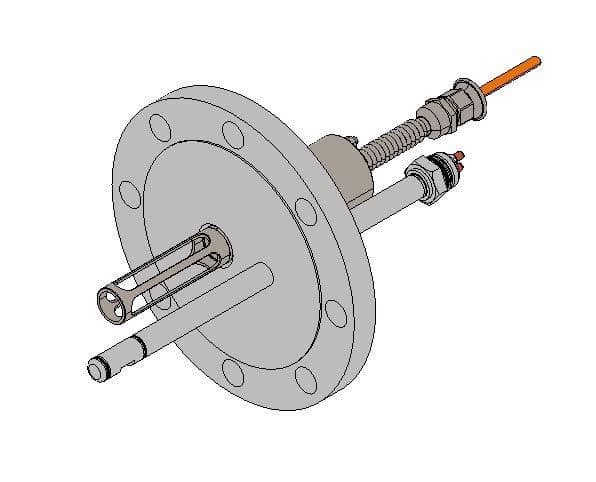

Image Credit: Guided Wave
By having both a NIRS measurement and a dynamic viscosity measurement; process engineers can make informed decisions based on real-time trends of polymer building, monomer reduction and weight-average molecular weight. Additionally, the dual probe configuration allows both measurements to occur under the same localized flow and sample conditions.
In other words, if the viscometer and NIRS probes were installed on separate flanges; in different locations on the reactor, then transient flow within the reactor could introduce a bias between the two techniques.
Overview of Polyurethane Production and the Role of PAT
Polyurethanes, like other similar step-growth polymerizations, are usually produced in two-step processes.
The first step of polymerization
Polymer materials of low average molecular weights (prepolymer); are produced through the reaction of a polyol and a large excess of diisocyanate; typically using a feed molar ration of 2:1. NIR Spectroscopy can measure the number of reactive hydroxyl groups (OH) on the polyol.
The OH number directly impacts the number of urethane linkages; which greatly influences the physical properties of the final polyurethane product. OH Number is, therefore, an important parameter to monitor and control during polyol production.
Additionally, the laboratory method that is commonplace for hydroxyl number determination is both time-consuming; and involves the use of hazardous materials. An In situ NIR transmission probe has faster throughput and reduce occupational exposure; to the hazardous materials required for offline testing.
The second step of polymerization
The polymer chain is extended; though the reaction of the prepolymer with a diol or polyol of low molecular weight (chain extender). Normally, the main objective is the production of polymer resins of large molecular weights at the end of the second reaction stp. To achieve this objective, some secondary objectives should be pursued.
First, monomer conversion and composition should be tightly controlled during the first reaction step. Second, the amount of polyol fed into the reaction vessel during the second step should be rigorously controlled.
These secondary control objectives are required to avoid monomer imbalance, which may lead to the production of polymer of low molecular weight and eventually cause the loss the batch product.
Finally, the evolution of weight average molecular weights should be accurately monitored by process viscometry and spectroscopy. The output of these process monitoring tools can then be used to control the weight average molecular weights and other parameters during the chain extension step of polyurethane synthesis.
Purposed Process Viscometry and Spectroscopy Control Scheme for Polyurethane Synthesis
The control logic shown below can be adapted as needed to meet the specific product requirements. During the first step, process spectroscopy is used to monitor the monomer conversion and weight average molecular weight.
Other parameters such as concentration or ratio of diisocyanates, excess water concentration, or ratio of glycol reaction products can also be measured with process spectroscopy.
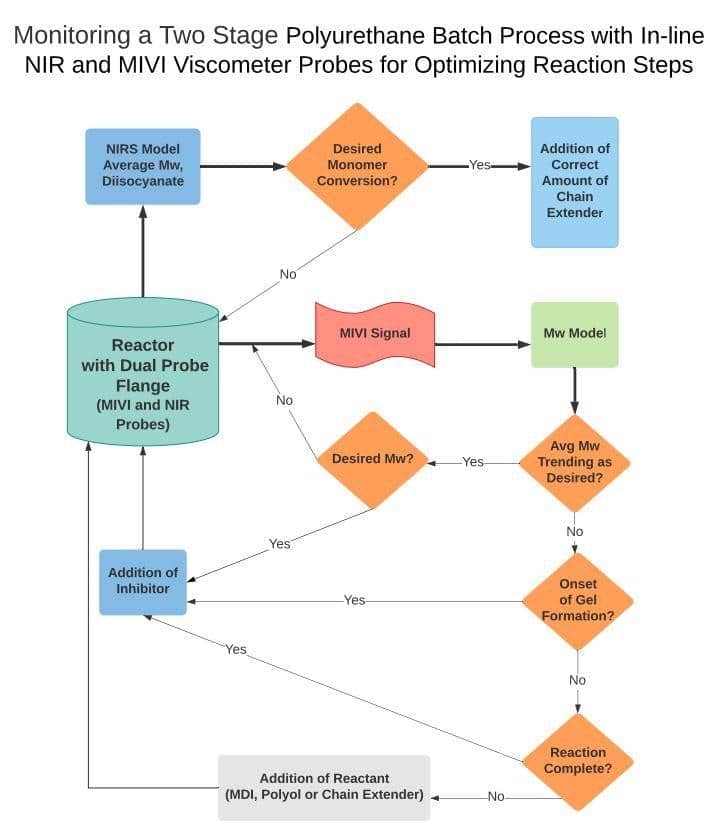
Image Credit: Guided Wave
During the second step of the reaction, the average polymer molecular weight is monitored by both process spectroscopy and viscometry. Depending on the trend of molecular weight, the reactant feed rate is adjusted or doses based on NIR and MIVI real-time data.
The dual measurement is an optimal mean to control the rate of polymerization towards an in-spec endpoint.
It should also be noted that the precision with which a NIR analyzer, such as the NIRO Full Spectrum Analyzer, can measure the average molecular weight begins to decrease as monomer conversion increases.
An adapted solution
A process viscometer such as the MIVI is therefore used to more precisely measure average molecular weight during the final stages of cross-linking and chain extension.
The onset of gelation can be determined by both process spectroscopy and viscometry. If the NIR determines that monomer conversion is occurring without a change in average molecular weight, then the onset of gelation has likely occurred.
Viscometry can be used as a secondary confirmation of gelation. If gelation is occurring, then the viscometer will measure a sudden perturbation dut to the agglomeration or branching of polymers.
The dual-probe approach enables the process engineer to quickly react to the gelation and begin to add inhibitors to slow the rate of cross-linking. The concentration of inhibitors, such as Hydrochloric Acid, can be measured by process spectroscopy.
Once the reaction trend has returned to normal, the amount of inhibitor can be reduced and the reactant, such as 1.4 Butane Diols, dosing feed rate to increase. The process control scheme allows the process engineer to coach the reaction to the desired molecular weight trajectories and ensure on spec product is produced.
Once the target molecular weight is achieved, the process engineer can transfer the on-spec polyurethane for any post-formation processing such as extrusion.
Conclusion
Bulk polymer production in and of itself is a fairly routine chemical process. However, the drive to optimize production and therefore reduce manufacturing costs necessitates the need for real-time process analysis.
Near-Infrared Process Spectroscopy and Viscometry; which includes the measurement of viscosity are both ideal candidates for determining the physical properties of polymers.
The development of modern process monitoring tools enables direct real-time measurements of the chemical reactions such as monomer conversion, molecular weight, acid number, or even the information of side branches and gels.
The dual probe and subsequent process control scheme can be used to improve batch copolymerization yield.
Designed by Elementale (Texas, USA) this probe was developed in collaboration with Guided Wave and Sofraser.
This information has been sourced, reviewed and adapted from materials provided by Guided Wave.
For more information on this source, please visit Guided Wave


